Project Optimus

Latest ICH GCP E6(R3) Amendment Explained
10 Reasons to Implement Modelling and Simulations Into Your Drug Development Program
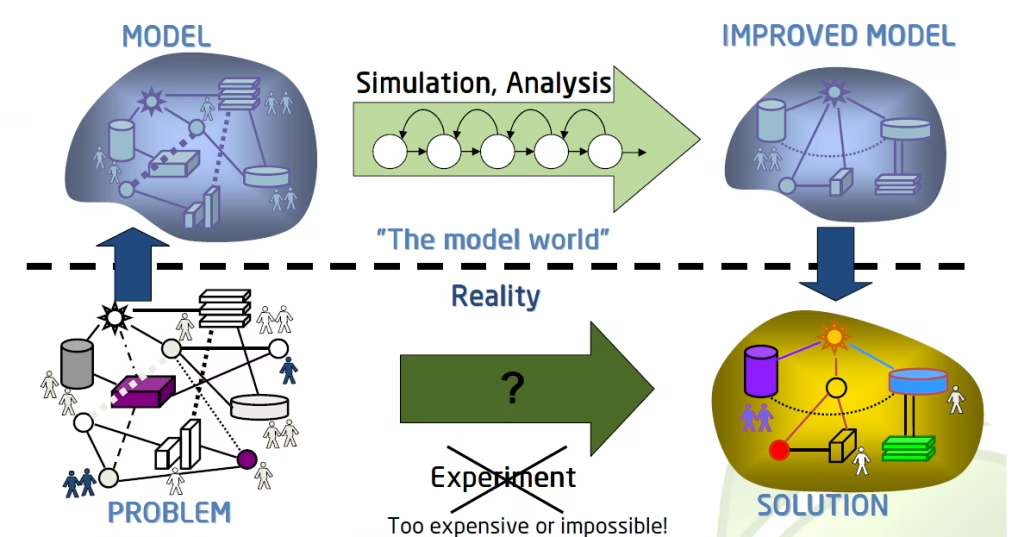
In the ever-evolving world of drug development, time and cost are crucial factors that can make or break a project. Traditional methods often involve lengthy trial-and-error processes, leading to high costs and delayed timelines. However, the integration of modelling and simulations offers a powerful solution. By leveraging advanced models, researchers can predict outcomes, optimize designs, and streamline development processes, all while reducing risks and costs.
In this article, we explore the 10 key reasons why incorporating modelling and simulations into your drug development program is a game-changer. From enhancing efficiency to improving scientific understanding, discover how these cutting-edge tools can transform your approach and increase the likelihood of success.
1. Cost Reduction
- Early Failure Identification: Models help identify potential drug failures early, reducing investment in unpromising candidates.
- Reduced Experimental Costs: Simulations can minimise reliance on costly clinical trial studies by narrowing down testable hypotheses.
2. Accelerated Development
- Faster Decision-Making: Predictive models enable quicker iterations and assessments of drug efficacy and safety.
- Optimisation of Trial Design: Simulations allow for efficient design of clinical trials, reducing time to market.
3. Enhanced Safety and Efficacy
- Risk Mitigation: Models can predict adverse events and refine dosing regimens to minimise risks.
- Individualised Treatment: Simulations can support the development of personalised medicine by accounting for variability in patient populations.
4. Increased Scientific Understanding
- Mechanistic Insights: Models help clarify biological pathways and mechanisms of action.
- Hypothesis Generation: Simulations can uncover new hypotheses for experimental validation.
5. Regulatory Support
- Regulatory Acceptance: Regulatory agencies increasingly recognise the value of modelling and simulations for supporting submissions and informing trial designs.
- Bridging Data Gaps: Models can extrapolate findings across populations or conditions, reducing the need for additional studies.
6. Reduction in Animal and Human Testing
- Ethical Benefits: Simulations reduce reliance on animal testing and potentially hazardous early-stage human trials.
7. Flexibility in Scenario Testing
- Exploration of Multiple Scenarios: Models allow researchers to test various conditions, dosages, and populations without real-world constraints.
- Stress Testing: Simulations can evaluate drug performance under extreme or rare conditions.
8. Improved Resource Allocation
- Prioritisation of Candidates: Simulations help rank drug candidates, focusing resources on the most promising ones.
- Streamlined Development Phases: Better-informed decisions can optimize resource use across development stages.
9. Support for Complex Systems
- Systems Pharmacology: Models integrate data across molecular, cellular, and system levels, offering holistic insights.
- Polypharmacy Assessment: Simulations can evaluate interactions in multi-drug regimens.
10. Learning and Continuous Improvement
- Data Integration: Models assimilate diverse datasets, enabling iterative refinement as new data becomes available.
- Knowledge Retention: Simulation frameworks preserve knowledge, facilitating reuse in future projects.
Integrating modelling and simulations into your drug development program can significantly streamline processes, reduce costs, and increase the likelihood of success. With the power to predict outcomes, optimise trial designs, and mitigate risks, the benefits are undeniable. Whether you’re aiming for cost efficiency, faster development, enhanced safety, or a deeper scientific understanding, the application of modelling and simulations can transform your approach.
To learn more about how our expertise can help accelerate your drug development program, contact us now and our team will guide you through every step, ensuring that you unlock the full potential of your drug candidates and streamline the drug development process.