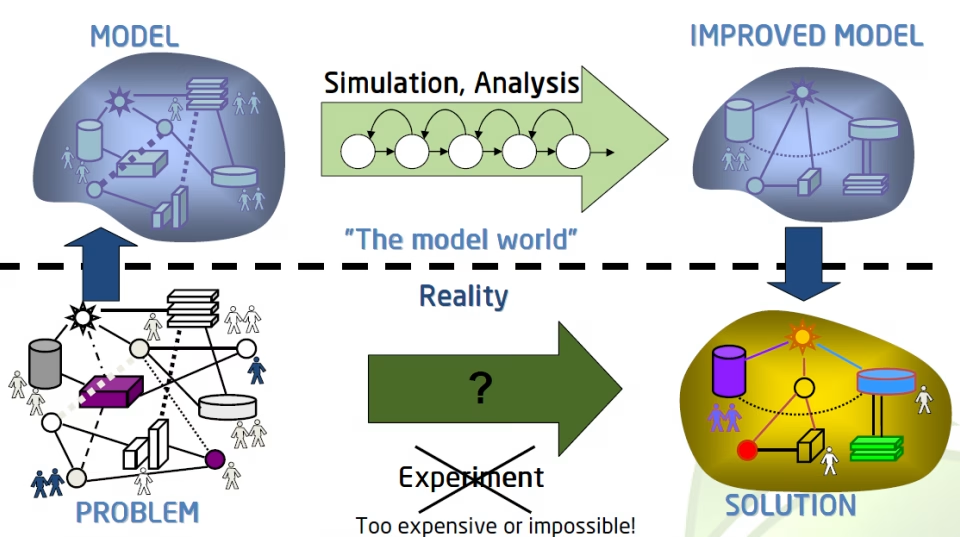
Overview A global biotech client sought to identify an efficacious human dose for a novel protein inhibitor prior to first-in-human (FIH) studies. Using a quantitative pharmacometrics […]
Latest ICH GCP E6(R3) Amendment Explained The International Council for Harmonisation (ICH) has unveiled a transformative update to its Good Clinical Practice (GCP) guidelines with the […]
10 Reasons to Implement Modelling and Simulations Into Your Drug Development Program In the ever-evolving world of drug development, time and cost are crucial factors that […]
What is Project Optimus? In 2016, the United States passed the 21st century Cure Act and along with that came the Cancer Moonshot initiative, which had […]
Pharmacokinetics in dermatology drug development Topical medications are applied to the skin or eyes to achieve a specific local therapeutic effect. However, delivering these medications is […]
Cannabis-based products for medical use in humans Guidance on the supply, manufacture, importation, and distribution of unlicensed cannabis-based products for medicinal use in humans (CBPMs) in […]