Latest ICH GCP E6(R3) Amendment Explained
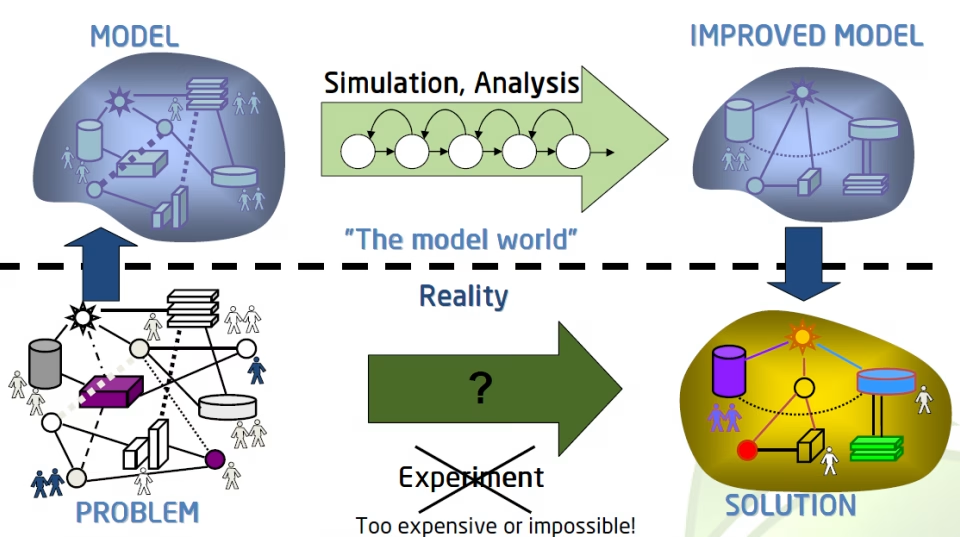
Overview
A global biotech client sought to identify an efficacious human dose for a novel protein inhibitor prior to first-in-human (FIH) studies. Using a quantitative pharmacometrics (PMx) approach, we integrated xenograft efficacy data, protein binding differences across species, and non-clinical PK/PD models to project human exposure and expected target inhibition.
Challenge
In mouse xenograft studies, the compound showed antitumor activity at 100 mg/kg BID, and the level of protein phosphorylation inhibition (%Pi) was closely correlated with plasma concentration (Cp).
However, protein binding varied substantially across species, making direct dose translation unreliable.
The client needed an evidence-based way to predict:
- Human pharmacokinetics
- Target engagement
- A clinically efficacious dose for FIH trials
Our Approach
We applied an integrated pharmacometrics (PMx) strategy:
1. Exposure–Response Modeling
We developed ER models linking Cp to %Pi based on the xenograft dataset.
2. Non-Clinical Population PK (PopPK) Modeling
A PopPK model was constructed using preclinical PK data to simulate human PK profiles.
3. Protein Binding Adjustment
Because protein binding differed across species, we corrected all Cp projections to human-equivalent free drug exposure, enabling a reliable estimate of the dose needed to achieve the target EC50.
4. Prediction of Human Efficacious Dose
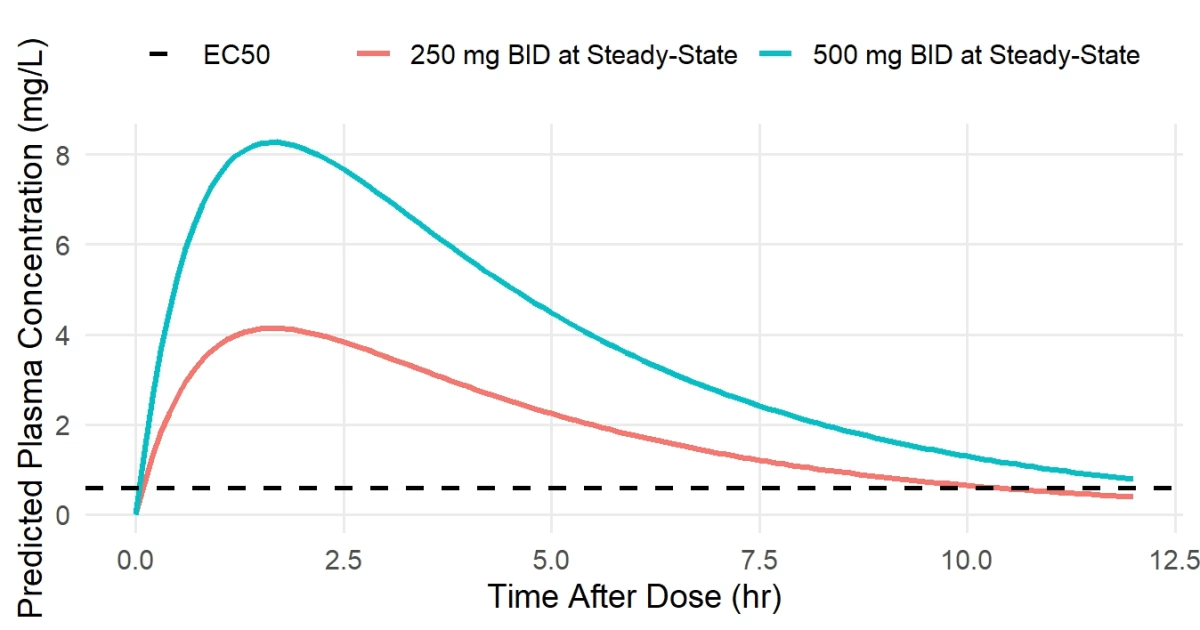
Simulations compared multiple BID regimens (e.g., 250 mg BID vs 500 mg BID) against the protein-binding-adjusted EC50 threshold.
Results
- Predicted human PK matched the observed data from the FIH study.
- Early clinical efficacy outcomes aligned with the PMx exposure–response predictions.
- The modeling framework provided confidence in dose selection and supported efficient progression into Phase 1.
A representative simulation figure is shown below, illustrating predicted steady-state concentrations and the target EC50 threshold for efficacy.
Impact
Our PMx-driven translational strategy enabled:
- A data-driven FIH dose selection
- Early confirmation of target engagement in humans
- Reduction in development risk through validated, mechanism-linked predictions
This case highlights the value of integrated PK/PD modeling in bridging non-clinical and clinical development.